JEE Main Important Physics formulas
ApplyAs per latest 2024 syllabus. Physics formulas, equations, & laws of class 11 & 12th chapters
NCERT Solutions for Class 9 Science Chapter 4 Structure of The Atom: After going through the structure of Atom class 9, you must be wondering that; what makes the atom of one element different from another element. Are atoms really indivisible, or are there smaller constituents inside the atom? With the assistance of CBSE NCERT solutions for Class 9 Science Chapter 4 Structure of the Atom, you will find out the answers to these questions. Through the NCERT solutions for the structure of atom chapter, you will also learn about sub-atomic particles and the various models that have been introduced to describe how these particles are arranged within the atom.
NCERT class 9 science chapter 4 question answer contains exercise-wise solutions that are created by the subject experts of Careers360. class 9 Chapter 4 Science, Structure of the Atom includes important topics and theories such as Thomson's model of an atom, Rutherford's model of an atom, Bohr's model of an atom, neutrons, etc. All these theories are very important for the exams. Apart from online solutions, the structure of atom class 9 pdf is also available, which students can download and use offline anytime, free of charge.
Free download class 9 science chapter 4 exercise question answer PDF for CBSE exam.
Topic 4.1 Charged particles in the matter?
Q. 1. What are canal rays?
Answer:
Canal rays are the positively charged radiations which consist of positively charged particles of atoms. they can pass through the perforated ( pierced ) cathode and then travel towards another cathode in a gas discharge tube.
They were given the name Canal rays by E. Goldstein in 1866 who discovered these radiations.
Q. 2. If an atom contains one electron and one proton, will it carry any charge or not?
Answer:
The proton is a positively charged particle and the electron is a negatively charged particle. their magnitude is equal and hence net charge in an atom is zero.
NCERT free solutions for class 9 science chapter 4 Structure of the Atom
Topic 4.2 The structure of an atom
Q. 1. On the basis of Thomson’s model of an atom, explain how the atom is neutral as a whole.
Answer:
According to Thomson’s model of an atom, an atom consists of a sphere of a positive charge.
The positive charge in the atom is spread all over like the red edible part of a watermelon, while the electrons are studded in the positively charged sphere, just like the seeds in the watermelon.
As negative and positive charges are equal in magnitude, they balance each other and thus the atom becomes electrically neutral as a whole.
Answer:
On the basis of Rutherford’s model of an atom, the subatomic particle which is present in the nucleus of an atom is Proton which is a positively charged particle.
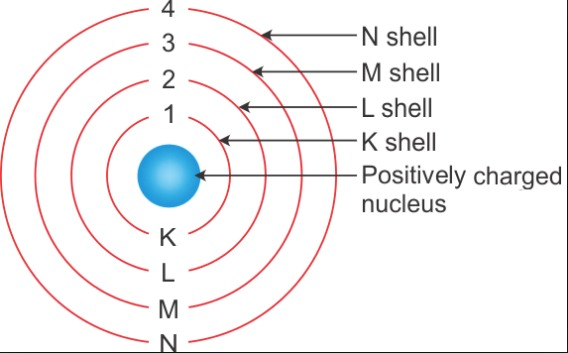
Q. 3. Draw a sketch of Bohr’s model of an atom with three shells.
Answer:
The sketch of Bohr’s model of an atom with three shells:
Answer:
If a foil of a heavy metal like platinum is used, then the observations in the alpha-particle scattering experiment would be the same as that in the gold foil experiment.
If a foil of a light metal like lithium is used, then the observations in the alpha-particle scattering experiment would not be the same because these metal are not so malleable so the thin foil is difficult to obtain.
The problem with not using thin foil is that the number of the alpha particle will bounce back from the thick foil and the location of positive mass would be difficult to find.
CBSE NCERT solutions for class 9 science chapter 4 Structure of the Atom
Topic 2.4.2 Neutron
Q. 1. Name the three sub-atomic particles of an atom.
Answer:
The three sub-atomic particles of an atom are :
1. Electron: a negatively charged particle
2. Proton: a positively charged particle
3. Neutron: a neutral particle
Answer:
The atomic mass of Helium = 4 u
No. of protons = 2
As atomic mass = no. of protons + no. of neutrons
No. of neutrons = At. mass - no. of protons
= 4 - 2
= 2
Hence Heium atom has 2 neutrons.
Solutions for NCERT class 9 science chapter 4 Structure of the Atom
Topic 4.3 How are electrons distributed in different orbits(shells)?
Q. 1. Write the distribution of electrons in carbon and sodium atoms.
Answer:
Number of electrons in carbon atom = 6
Number of electrons in sodium atom = 11
Electron Distribution:
| Element | First Orbit or K -shell | Second Orbit or L-shell | Third Orbit or M-shell |
| Carbon | 2 | 4 | 0 |
| Sodium | 2 | 8 | 1 |
Q. 2. If and
shells of an atom are full, then what would be the total number of electrons in the atom?
Answer:
Maximum Number of electrons in K-shell = 2
Maximum Number of electrons in L-shell = 8
The total no. of electrons in the atom = 2 + 8
= 10
If and
shells of an atom are full, then the total number of electrons in the atom will be 10.
NCERT solutions for class 9 science chapter 4 Structure of the Atom
Topic 4.4 Valency
Q. 1. How will you find the valency of chlorine, sulphur and magnesium?
Answer:
Valancy is basically the minimum number of the electron we have to add or remove such that every shell in the atom is completely filled.
Mathematically,
when the outermost shell of an atom contains 4 or less than 4 electrons, its valency is equal to the number of valence electrons in the outermost shell and when the outermost shell contains more than 4 electrons, the valency of the atom is equal to 8 - no. of valence electrons in the atom.
Chlorine :
Atomic No. of Cl = 17
Its electronic configuration = 2, 8, 7
Valency of Cl = 8 - 7 = 1
Sulphur :
Atomic no. of S = 16
Its electronic configuration = 2, 8, 6
Valency of S = 8 - 6 = 2
Magnesium:
Atomic no. of Mg = 12
Its electronic configuration = 2, 8, 2
Valency of Mg = 2
NCERT textbook solutions for class 9 science chapter 4 Structure of the Atom
Topic 4.5 Atomic number and Mass number
Q. 1.(i) If the number of electrons in an atom is 8 and the number of protons is also 8, then
(i) what is the atomic number of the atom?
Answer:
Given,
Number of electrons in the atom = 8
Number of proton in the atom = 8
The atomic number of an atom is equal to the number of proton in that atom. hence the atomic number of the given atom is 8.
Q. 1. (ii) If the number of electrons in an atom is 8 and the number of protons is also 8, then
(ii) what is the charge on the atom?
Answer:
In the given atom, the total number of positive charges is equal to the total number of negative charge.
Number of Protons (8) = Number of electrons (8)
They both will neutralize each other. So, the atom will not possess any charge.
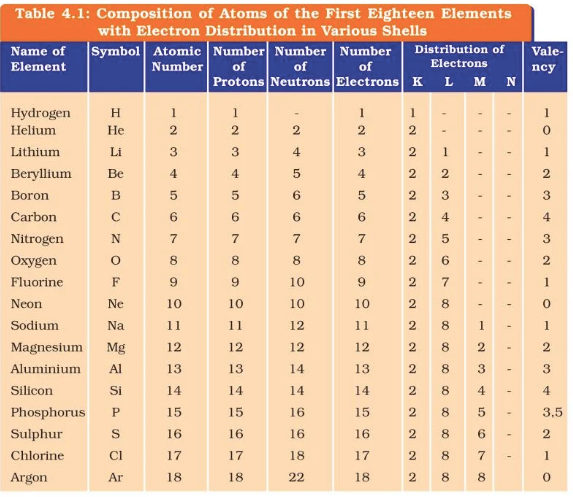
Q. 2. With the help of Table 4.1, find out the mass number of oxygen and sulfur atom.
Answer:
For Oxygen:
Number of protons = 8
Number of electrons = 8
Mass number = Number of Protons + Number of neutrons
= 8 + 8
= 16
Hence mass number for Oxygen is 16.
For Sulphur:
Number of protons = 16
Number of electrons = 16
Mass number = Number of Protons + Number of neutrons
= 16 + 16
= 32
Hence Mass number for Sulphur is 32.
CBSE NCERT solutions for class 9 science chapter 4 Structure of the Atom
Topic 4.6 Isotopes
Q. 1. For the symbol and
tabulate three sub-atomic particles found in each of them.
Answer:
H, D, and T are the three isotopes of hydrogen with the same atomic number and different mass numbers of 1, 2 and 3 respectively.
| Element | Symbol | Number of Electrons | Number of Protons | Number of Neutrons |
| Hydrogen | H | 1 | 1 | 0 |
| Deuterium | D | 1 | 1 | 1 |
| Tritium | T | 1 | 1 | 2 |
Q. 2. Write the electronic configuration of any one pair of isotopes and isobars.
Answer:
Isotopes :
Isotopes are the atoms with the same number of proton and different atomic mass. The difference in atomic mass arises due to the different number of neutrons present in the atom.
Some Examples of Isotopes are :
1. and
,
2. and
.
Isobar:
Isobars are the atom with the same atomic mass and different atomic number.
Some example of Isobars are :
1. and
2. and
Q. 1. Compare the properties of electrons, protons, and neutrons.
Answer:
The Comparison of Properties between Electron, Proton, and Neutron:
| Properties | Electrons | Protons | Neutrons |
| Charge | Negatively charged | Positively charged | No charge |
| Weight | Negligible | 1 a.m.u | 1 a.m.u |
| Location in atom | Outside the nucleus | Inside the nucleus | Inside the nucleus |
| Reaction with a charged particle | Attracts positive charge | Attracts negative charge | gives no reaction to any charge |
Q. 2. What are the limitations of J.J. Thomson’s model of the atom?
Answer:
The limitations of J.J. Thomson’s model of the atom are:
1. Thomson's model of the atom could not explain the results of alpha particle scattering experiment carried out by Rutherford. this model failed to depict why most of the alpha particle passes through gold foil and why some of them got diverted in different angles and some of them rebounded and returned back to their paths.
2. It was solely based on the imagination and did not have any experimental evidence.
Q. 3. What are the limitations of Rutherford’s model of the atom?
Answer:
The limitations of Rutherford's model of the atom is that It does not explain the stability of the atom. As we know now, when charged bodies move in a circular motion, they emit radiations.
This means that the electrons revolving around the nucleus (as suggested by Rutherford) would lose energy and come closer and closer to the nucleus, and a stage will come when they would finally merge into the nucleus.
This makes the atom unstable, which is clearly not the case. The electrons do not fall into the nucleus, atoms are very stable and do not collapse on their own.
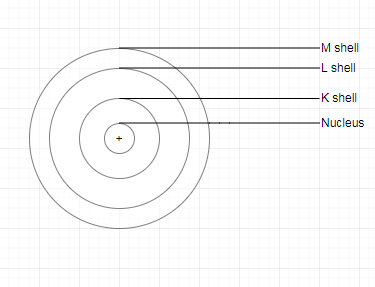
Q. 4. Describe Bohr’s model of the atom.
Answer:
In order to overcome the objections raised against Rutherford's model of the atom, Neils Bohr put forward his model of the atom. According to Bohr's model of the atom,
1. An atom holds the nucleus in the center. the whole mass of the atom is concentrated at the nucleus.
2. The negatively charged particle revolves around the nucleus in definite circular paths known as orbits or which are designated as K, L, M, N, etc. or numbered as n = 1, 2, 3, 4, etc. (outward from the nucleus).
3. While revolving in discrete orbits, the electrons do not radiate energy. But when an electron jumps from one energy level to another, the energy of the atom changes.
Q. 5. Compare all the proposed models of an atom given in this chapter.
Answer:
Comparison of different proposed Model:
| Feature | Thomson's Model | Rutherford's Model | Bohr's Model |
| Positive Charge | The Positive charge is distributed in Sphere | The positive charge is concentrated at the core of the atom, which is called the nucleus | The positive charge is present in the core of the atom, called nucleus. |
| Negative Charge | The electrons are embedded in the positively charged sphere of an atom, like the seeds in a watermelon. | The nucleus is surrounded by electrons, and the electrons and the nucleus are held together by the electrostatic force of attraction | The electrons move in discrete orbits, and each orbit is associated with a definite amount of energy. |
| Limitation | This model could not explain the results of an alpha particle scattering experiment | This model could not explain the stability of the atom. | This model perfectly explains the stability of an atom |
| Diagrammatic representation |  | 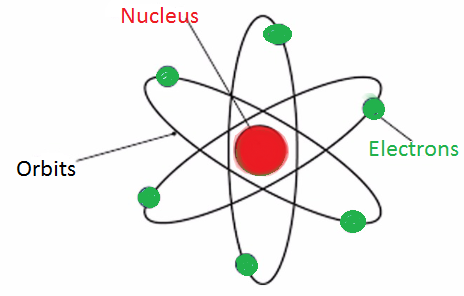 | 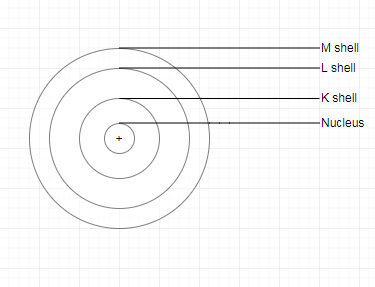 |
Answer:
The Bohr and Bury scheme for the distribution of electrons in an atom is based on the following rules :
1. The maximum number of electrons that a shell can have is represented by , where n is the quantum number of that particular energy shell. Thus, the maximum number of electrons in the first four shells are :
1st (K) shell 2 x = 2
2nd (L) shell 2 x = 8
3rd (M) shell 2 x = 18
4th (N) shell 2 x = 32
2. The outermost shell, which is also called valence shell, can have a maximum of 8 electrons.
3. If permitted by rule 1, The shell inner to the outermost shell (the second last shell ) can accommodate a maximum of 18 electrons.
4. Electrons are not taken in unless the inner shells are filled, i.e., the shells are filled in a step-wise manner.
Q. 7. Define valency by taking examples of silicon and oxygen.
Answer:
The definite combining capacity of an atom of an element, in which electrons are lost, gained or shared with other atoms to complete the octave in the outermost shell is defined as valency.
In other words,
Valancy is basically the minimum number of the electron we have to add or remove from or in the outermost shell such that every shell in the atom is completely filled.
And Mathematically,
when the outermost shell of an atom contains 4 or less than 4 electrons, its valency is equal to the number of valence electrons in the outermost shell and when the outermost shell contains more than 4 electrons, the valency of the atom is equal to 8 - no. of valence electrons in the atom.
The valency of Silicon:
Atomic number = 14
Distribution of electron :
K = 2
L = 8
M = 4
Number of electrons in outermost shell = 4
Valency = 8 - 4 = 4.
The valency of Oxygen:
Atomic number = 8
Distribution of electrons:
K = 2
L = 6
The number of electron in outermost shell = 6
Valency = 8 - 6 = 2
Q. 8.(i) Explain with examples
(i) Atomic number,
Answer:
Atomic Number :
An atomic number of an atom is the total number of protons present within the nucleus of an atom is known as the atomic number. it is denoted by symbol Z.
Example: As the Oxygen atom has 8 protons in its nucleus, its atomic number is 8.
Q. 8.(ii) Explain with examples
(ii) Mass number,
Answer:
Mass Number :
The mass number of an atom is the sum total of the masses of all the nucleons present in the nucleus of an atom, i.e.,
Mass Number = No. of Protons + No. of Neutrons
It is denoted by A.
Example: As a sodium atom has 11 protons and 12 neutrons in its nucleus,
So, it's mass number = 11 + 12 = 23.
Q. 8.(iii) Explain with examples
(iii) Isotopes
Answer:
Isotopes:
Isotopes are the atoms of the same element having the same atomic number but a different mass number.
Example: Carbon molecule exists as and
.
Q. 8.(iv) Explain with examples
(iv) Isobars.
Answer:
Isobar:
Isobars are the atoms of different elements having the same mass number but different atomic numbers.
Example: and
. Mass numbers of calcium and argon atoms have different atomic numbers (20 and 18 ) but the same mass number 40.
Two uses of isotopes are:
(i) An isotope of uranium is used as fuel in nuclear reactors.
(ii) An isotope of cobalt is used in the treatment of cancer.
Q. 9. has completely filled
and
shells. Explain.
Answer:
The atomic number of Na = 11
No. of electrons in Na atom = 11
In ,the positive charge is obtained due to the loss of one electron from the M shell of Na atom.
So, No. of electrons in Na + ion = 11 -1 = 10
Hence, electronic configuration of Na + = 2, 8
In Na + , K and L shells are completely filled since K shell can have a maximum of 2 electrons and L shell can have a maximum of 8 electrons.
Answer:
Given, two isotopes and
.
Average atomic mass:
.
Answer:
Given, the average atomic mass of a sample of an element is
.
Two isotopes of element = and
Now, Let's percent of isotope be x and percent of
be 100 - x
So, According to the question,
Average Atomic Mass :
Hence the percentage of isotope is 90 % and thepercentage isotope
is 10%.
Q. 12. If , what would be the valency of the element? Also, name the element.
Answer:
Given
the Atomic number, Z = 3
Distribution of electrons :
K = 2,
L = 1
So, Valency = 1 .
The element with atomic number 3 is lithium.
Q. 13. Composition of the nuclei of two atomic species and
are given as under
X Y
Protons = 6 6
Neutrons = 6 8
Give the mass numbers of and
. What is the relation between the two species?
Answer:
As we know,
the mass number of an atom = No. of protons + No. of Neutrons
So,
The mass number of X = No. of protons of X + No. of Neutrons of X
= 6 + 6
= 12
The mass number of Y = No. of protons of Y + No. of Neutrons of Y
= 6 + 8
= 14
As both X and Y have the same atomic number (6) but different numbers (i.e., 12 and 14 respectively), they are isotopes.
Q. 14. For the following statements, write T for True and F for False.
(a) J.J. Thomson proposed that the nucleus of an atom contains only nucleons.
(b) A neutron is formed by an electron and a proton combining together. Therefore, it is neutral.
(c) The mass of an electron is about 1 2000 times that of a proton.
(d) An isotope of iodine is used for making tincture iodine, which is used as a medicine.
Put a tick against correct choice and cross (×) against wrong choice in questions 15, 16 and 17
Answer:
(a) The statement is False.
(b) The statement is False.
(c) The statement is True.
(d) The statement is True.
Q. 15. Rutherford’s alpha-particle scattering experiment was responsible for the discovery of
(a) Atomic Nucleus
(b) Electron
(c) Proton
(d) Neutron
Answer:
Rutherford’s alpha-particle scattering experiment was responsible for the discovery of the Atomic Nucleus.
Hence, option (a) is the correct answer.
Q. 16. Isotopes of an element have
(a) the same physical properties
(b) different chemical properties
(c) different number of neutrons
(d) different atomic numbers
Answer:
Isotopes of an element have a different number of neutrons.
Hence option (c) is correct.
Q. 17. A Number of valence electrons in ion are:
(a) 16
(b) 8
(c) 17
(d) 18
Answer:
The Electronic configuration of ion is :
K = 2
L = 8
M = 8
Hence Number of valance electron in ion = 8.
Hence, option (b) is the correct answer.
Q. 18. Which one of the following is a correct electronic configuration of sodium?
(a) 2,8
(b) 8,2,1
(c) 2,1,8
(d) 2,8,1.
Answer:
The atomic number of sodium = 11
The electronic configuration of the sodium :
K = 2
L = 8
M = 1
Hence, option (d) is correct.
Q. 19. Complete the following table.
Answer:
| Atomic Number | Mass Number | Number of Neutrons | Number of Protons | Number of Electrons | Number of Atomic Species |
| 9 | - | 10 | - | - | - |
| 16 | 32 | - | - | - | Sulphur |
| - | 24 | - | 12 | - | - |
| - | 2 | - | 1 | - | - |
| - | 1 | 0 | 1 | 0 | - |
First row:
atomic number = 9
so, the element is Fluorine.
Atomic no. = No. of protons = no. of electrons = 9
Mass number = no. of protons + no. of neutrons = 9 + 10 = 19
Second row:
Since atomic no. is 16 so, no. of protons = no. of electrons = 16
No. of neutrons = Mass no. - no. of protons = 32 - 16 = 16
Third row:
No. of protons = Atomic no. = 12
So, the element is Magnesium.
No. of electrons = no. of protons = 12
No. of neutrons = Mass no. - no. of protons = 24 - 12 = 12
Fourth row:
No. of protons = Atomic no. = 1
So, the element is Deuterium.
No. of electrons = no. of protons = 1
No. of neutrons = Mass no. - no. of protons = 2 - 1 = 1
Fifth row:
No. of protons = Atomic no. = 1
The element is Protium since the mass number is 1.
So the Table becomes,
| Atomic number | mass number | Number of neutrons | Number of protons | Number of electrons | Name of the element |
| 9 | 19 | 10 | 9 | 9 | Fluorine |
| 16 | 32 | 16 | 16 | 16 | Sulfur |
| 12 | 24 | 12 | 12 | 12 | Magnesium |
| 1 | 2 | 1 | 1 | 1 | Deuterium |
| 1 | 1 | 1 | 1 | 0 | Hydrogen ion |
| Chapter No. | Chapter Name |
| Chapter 1 | Matter in Our Surroundings |
| Chapter 2 | Is Matter Around Us Pure |
| Chapter 3 | Atoms and Molecules |
| Chapter 4 | Structure of The Atom |
| Chapter 5 | The Fundamental Unit of Life |
| Chapter 6 | Tissues |
| Chapter 7 | Diversity in Living Organisms |
| Chapter 8 | Motion |
| Chapter 9 | Force and Laws of Motion |
| Chapter 10 | Gravitation |
| Chapter 11 | Work and Energy |
| Chapter 12 | Sound |
| Chapter 13 | Why Do We Fall ill? |
| Chapter 14 | Natural Resources |
| Chapter 15 | Improvement in Food Resources |
Formulas are essential tools for solving numerical problems and gaining a detailed understanding of a chapter. Here are some important formulas for the science chapter 4 class 9, which will enable you to solve questions smoothly and efficiently.
Maximum number of electrons in different shells
According to the Bohr model of the atom, the maximum number of electrons that can occupy a shell is given by the formula 2n2, where n represents the shell number. The first shell (n = 1) can hold a maximum of 2 electrons, the second shell (n = 2) can hold a maximum of 8 electrons, and so on.
Average atomic mass
Average atomic mass = (M1× P1) + (M2 × P2) + ... + (Mn × Pn)
Where:
M1, M2 ..., and Mn represent the individual atomic masses of the isotopes of the element.
P1, P2 ..., and Pn represent their respective natural abundances as decimal fractions or percentages (converted to decimal form).
For a comprehensive collection of class 9 science chapter-wise formulas, a downloadable handbook is available at the provided link. Students can utilize this resource to access formulas for each chapter, aiding them in solving questions, revising concepts, and collaborating with their peers effectively. This handbook serves as a valuable tool for enhancing understanding and achieving success in science examinations.
The important topics for class 9 chapter 4 science, "Structure of the Atom," are highly significant for exams and school tests. By focusing on these key areas during revision, students can efficiently review the chapter and consolidate their understanding. Here are the important topics for quick revision:
Also Check -
NCERT Books and NCERT Syllabus :
| Chapter No. | Chapter Name |
| Chapter 1 | Chapter 1 Matter in our Surroundings |
| Chapter 2 | Chapter 2 Is Matter Around Us Pure? |
| Chapter 3 | Chapter 3 Atoms and Molecules |
| Chapter 4 | Chapter 4 Structure of the Atom |
| Chapter 5 | Chapter 5 The Fundamental Unit of Life |
| Chapter 6 | Chapter 6 Tissues |
| Chapter 7 | Chapter 7 Diversity in Living Organisms |
| Chapter 8 | Chapter 8 Motion |
| Chapter 9 | Chapter 9 Forces and Laws of Motion |
| Chapter 10 | Chapter 10 Gravitation |
| Chapter 11 | Chapter 11 Work and Energy |
| Chapter 12 | Chapter 12 Sound |
| Chapter 13 | Chapter 13 Why do We Fall ill? |
| Chapter 14 | Chapter 14 Natural Resources |
| Chapter 15 | Chapter 15 Improvement in Food Resources |
The main topics covered in NCERT text book for Class 9 Science chapter 4 are-
The main components of an atom are protons, neutrons, and electrons. Protons and neutrons are located in the nucleus, while electrons orbit around the nucleus.
Isotopes are atoms of the same element that have the same number of protons but differ in the number of neutrons. This results in isotopes having different mass numbers.
The chapter discusses several atomic models, including Thomson's plum pudding model, Rutherford's nuclear model, and Bohr's model of the atom.
To revise the chapter effectively, focus on understanding the key concepts, practice numerical problems, and review the important formulas related to atomic structure. Additionally, refer to the NCERT textbook, study guides, and solved examples for comprehensive preparation.

As per latest 2024 syllabus. Physics formulas, equations, & laws of class 11 & 12th chapters

Ranked #52 Among Universities in India by NIRF | Up to 30% Merit-based Scholarships | Lifetime placement assistance

Get up to 90% scholarship on NEET, JEE & Foundation courses

As per latest 2024 syllabus. Chemistry formulas, equations, & laws of class 11 & 12th chapters

Enrol in PACE IIT & Medical, Financial District, Hyd for JEE/NEET preparation

Start your JEE preparation with ALLEN