-
Engineering and Architecture
Exams
Colleges
Predictors
Resources
-
Computer Application and IT
Quick Links
Colleges
-
Pharmacy
Colleges
Resources
-
Hospitality and Tourism
Colleges
Resources
Diploma Colleges
-
Competition
Other Exams
Resources
-
School
Exams
Ranking
Products & Resources
-
Study Abroad
Top Countries
Student Visas
-
Arts, Commerce & Sciences
Exams
Colleges
Upcoming Events
Resources
-
Management and Business Administration
Exams
Colleges & Courses
Predictors
-
Learn
Online Courses
Engineering Preparation
Medical Preparation
-
Online Courses and Certifications
Top Streams
Specializations
- Digital Marketing Certification Courses
- Cyber Security Certification Courses
- Artificial Intelligence Certification Courses
- Business Analytics Certification Courses
- Data Science Certification Courses
- Cloud Computing Certification Courses
- Machine Learning Certification Courses
- View All Certification Courses
Resources
-
Medicine and Allied Sciences
Colleges
Predictors
Resources
-
Law
Resources
Colleges
-
Animation and Design
Animation Courses
Colleges
Resources
-
Media, Mass Communication and Journalism
Exams
Colleges
Resources
-
Finance & Accounts
Top Courses & Careers
Colleges
Nature of C-X bond and Physical Properties - (Concept)
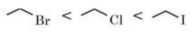
Halogen atoms are more electronegative than carbon, therefore, carbon-halogen bond of alkyl halide is polarised; the carbon atom bears a partial positive charge whereas the halogen atom bears a partial negative charge. As we go down the group in the periodic table, the size of halogen atom increases. Fluorine atom is the smallest and iodine atom is the largest. Consequently, the carbon-halogen bond length also increases from to
.

Physical properties of Haloalkanes and Haloarenes:
(1) These are less soluble in but more soluble in Organic solvents
(2) Their density follows the order:
Iodide > Bromide > Fluoride > Chloride
(3) Their boiling point follows the order:
Iodide > Bromide > Fluoride > Chloride
(4) Boiling point of Isomeric haloalkanes decreases with the increase in branching
(5) Boiling point of isomeric dihalobenzene is nearly the same and follows the order
para > ortho > meta
| Exam | Chapter |
| JEE MAIN | Organic Compounds containing Halogens |
Which of the following are monohalides?

| A. |
|
| B. |
|
| C. |
|
| D. |
|
Which ones are dihalogen derivatives of alkanes?

| A. |
|
| B. |
|
| C. |
|
| D. |
|
Which of the following are geminal dihalides?
a)
b)
c)
| A. |
|
| B. |
|
| C. |
|
| D. |
|
Which of the following are vicinal dihalides?

| A. |
|
| B. |
|
| C. |
|
| D. |
|
Match the following trihalogen derivatives to their use
| Reagent | Use |
| (a) Chloroform | (p) Disinfectant |
| (b) Bromoform | (q) Anesthetic |
| (c) Iodoform | (r) Laboratory Reagent |
| A. |
|
| B. |
|
| C. |
|
| D. |
|
Which ones are tetrahalogen derivatives?
(a) (b)
(c)
| A. |
|
| B. |
|
| C. |
|
| D. |
|
The major product formed in the following reaction is :

| A. |
|
| B. |
|
| C. |
|
| D. |
|
Match the following correctly
|
(a) |
(p) Geminal dihalide |
|
(b) |
(q) Tertiary alkylhalide |
|
(c) |
(r) Vicinal dihalide |
|
(d) |
(s) Chloroform |
| A. |
|
| B. |
|
| C. |
|
| D. |
|
Which of the followinng compounds are vinyl chlorides?
a)
b)

| A. |
|
| B. |
|
| C. |
|
| D. |
|
An 'Assertion' and a 'Reason' are given below. Choose the correct answer from the following options:
Assertion (A): Vinyl halides do not undergo nucleophilic substitution easily.
Reason (R): Even though the intermediate carbocation is stabilized by loosely held electrons, the cleavage is difficult because of strong bonding.
| A. |
|
| B. |
|
| C. |
|
| D. |
|
Which of the following is benzylic halide?
| A. |
|
| B. |
|
| C. |
|
| D. |
|
Which of the following is an allyl halide?
| A. |
|
| B. |
|
| C. |
|
| D. |
|
Which of the following will most readily give the dehydrohalogenation product ?
| A. |
|
| B. |
|
| C. |
|
| D. |
|
Which of the following can be classified as haloalkanes?
a)
b)
c)
d)
| A. |
|
| B. |
|
| C. |
|
| D. |
|
The correct decreasing order of densities of the following compounds is :
| A. |
|
| B. |
|
| C. |
|
| D. |
|
The absolute configuration of

is :
| A. |
|
| B. |
|
| C. |
|
| D. |
|
Arrange the following in the correct order of dipole moment.
| A. |
|
| B. |
|
| C. |
|
| D. |
|
Arrange the following in the correct order of decreasing bond enthalpy
| A. |
|
| B. |
|
| C. |
|
| D. |
|
Identify the correct order for the given property for following compounds.
(A) Boiling Point:
(B) Density:
(C) Boiling Point:
(D) Density:
(E) Boiling Point:
Choose the correct answer from the option given below:
| A. |
|
| B. |
|
| C. |
|
| D. |
|
The correct order of melting points of dichlorobenzenes is
| A. |
|
| B. |
|
| C. |
|
| D. |
|

Since halogen atoms are more electronegative than carbon, the carbonhalogen bond of alkyl halide is polarised; the carbon atom bears a partial positive charge whereas the halogen atom bears a partial negative charge.