-
Engineering and Architecture
Exams
Colleges
Predictors
Resources
-
Computer Application and IT
Quick Links
Colleges
-
Pharmacy
Colleges
Resources
-
Hospitality and Tourism
Colleges
Resources
Diploma Colleges
-
Competition
Other Exams
Resources
-
School
Exams
Ranking
Products & Resources
-
Study Abroad
Top Countries
Student Visas
-
Arts, Commerce & Sciences
Exams
Colleges
Upcoming Events
Resources
-
Management and Business Administration
Exams
Colleges & Courses
Predictors
-
Learn
Online Courses
Engineering Preparation
Medical Preparation
-
Online Courses and Certifications
Top Streams
Specializations
- Digital Marketing Certification Courses
- Cyber Security Certification Courses
- Artificial Intelligence Certification Courses
- Business Analytics Certification Courses
- Data Science Certification Courses
- Cloud Computing Certification Courses
- Machine Learning Certification Courses
- View All Certification Courses
Resources
-
Medicine and Allied Sciences
Colleges
Predictors
Resources
-
Law
Resources
Colleges
-
Animation and Design
Animation Courses
Colleges
Resources
-
Media, Mass Communication and Journalism
Exams
Colleges
Resources
-
Finance & Accounts
Top Courses & Careers
Colleges
Which one of the following orders is correct for the bond dissociation enthalpy of halogen molecules?
Following are the two statements regarding the origin of life :
- The earliest organisms that appeared on the earth were non-green and presumably anaerobes.
- The first autotrophic organisms were the chemoautotrophs that never released oxygen.
Of the above statements which one of the following options is correct?
(a) is correct but (b) is false.
(b) is correct but (a) is false.
Both (a) and (b) are correct.
Both (a) and (b) are false.
and
both when dissolved in water containing I- ions the pair of species formed is:
The angular momentum of electron in 'd' orbital is equal to:
Transfer characteristics [output voltage (V0) vs input voltage (Vi)] for a base biased transistor in CE configuration is as shown in the figure. For using transistor as a switch, it is used:
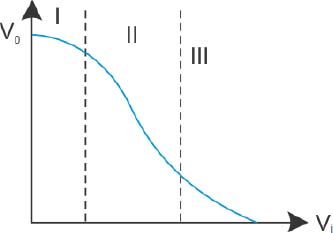
In region III
Both in region (I) and (III)
In region II
In region I
Which statement is wrong for viruses
All are parasites
All of them have helical symmetry
They have ability ot synthesize nucleic acids and proteins
Antibiotics have no effect on them
Kyoto Protocol was endorsed at:
CoP-4
CoP-3
CoP-5
CoP-6
The tendency of population to remain in genetic equilibrium may be disturbed by:
lack of random mating
random mating
lack of migration
lack of mutations
Select the correct match of the digested products in humans given in column I with their absorption site and mechanism in column II.
|
Column I |
Column II |
|
Cholesterol, maltose |
large intestine, active absorption |
|
Glycine, glucose |
small intestine, active absorption |
|
Fructose, Na+ |
small intestine, passive absorption |
|
Glycerol, fatty acids |
duodenum, move as chilomicrons |
The Air Prevention and Control of Pollution Act came into force in:
1990
1975
1981
1985
A technique of micropropagation is:
Protoplast fusion
Embryo rescue
Somatic hybridization
Somatic embryogenesis
A population will not exist in Hardy-Weinberg equilibrium if :
there is no migration
the population is large
individuals mate selectively
there are no mutations
The internal energy change in a system that has absorbed 2 Kcals of heat and done 500 J of work is:
6400 J
5400 J
7900 J
8900 J
The mean free path of electrons in a metal is 4 x 10-8 m. The electric field which can given on an average 2 eV energy to an electron in the metal will be in units of V/m:
5 x 10-11
8 x 10—11
5 x 107
8 x 107
Under the influence of a uniform magnetic field, a charged particle moves with constant speed V in a circle of radius R. The time period of rotation of the particle:
depends on R and not on V
is independent of both V and R
depends on both V and R
depends on V and not on R
In a standard ECG which one of the following alphabets is the correct representation of the respective activity of the human heart?
S - start of systole
T - end of diastole
P - depolarisation of the atria
R - repolarisation of ventricles
DDT residues are rapidly passed through food chain causing biomagnification because DDT is:
moderately toxic
non-toxic to aquatic animals
water soluble
lipo soluble
Middle lamella is composed mainly of:
Muramic acid
Calcium pectate
Phosphoglycerides
Hemicellulose
The energy absorbed by each molecule (A2) of a substance is 4.4 x 10-19 J and bond energy per molecule is 4.0 x 10-19 J. The kinetic energy of the molecule per atom will be:
2.2 10-19 J
2.0 10-19 J
4.0 10-20 J
2.0 10-20 J
Chipko movement was launched for the protection of
forests
livestock
wet lands
grasslands
One of the commonly used plant growth hormone in tea plantations is
Ethylene
Abscisic acid
Zeatin
Indole - 3 - acetic acid
The displacement of a particle along the x-axis is given by x = asin2t. The motion of the particle corresponds to
Simple harmonic motion of frequency /2
Simple harmonic motion of frequency /
Simple harmonic motion of frequency 3/2
Non simple harmonic motion
The correct order of increasing bond angles in the following species is :
< Cl2O < ClO2
Cl2O < ClO2 <
ClO2 < Cl2O <
Cl2O < < ClO2
Which one of the following kinds of animals are triploblastic?
Corals
Flat worms
Sponges
Ctenophores
A solution contains Fe2+, Fe3+ and I- ions. This solution was treated with iodine at 35°C. Eo for Fe3+/Fe2+ is +0.77 V and Eo for .The favourable redox reaction is :
I- will be oxidised to I2
Fe2+ will be oxidised to Fe3+
I2 will be reduced to I-
There will be no redox reaction

