-
Engineering and Architecture
Exams
Colleges
Predictors
Resources
-
Computer Application and IT
Quick Links
Colleges
-
Pharmacy
Colleges
Resources
-
Hospitality and Tourism
Colleges
Resources
Diploma Colleges
-
Competition
Other Exams
Resources
-
School
Exams
Top Schools
Products & Resources
-
Study Abroad
Top Countries
Resources
-
Arts, Commerce & Sciences
Colleges
Upcoming Events
Resources
-
Management and Business Administration
Exams
Colleges & Courses
Predictors
-
Learn
Law Preparation
MBA Preparation
Engineering Preparation
Medical Preparation
-
Online Courses and Certifications
Top Streams
Specializations
- Digital Marketing Certification Courses
- Cyber Security Certification Courses
- Artificial Intelligence Certification Courses
- Business Analytics Certification Courses
- Data Science Certification Courses
- Cloud Computing Certification Courses
- Machine Learning Certification Courses
- View All Certification Courses
Resources
-
Medicine and Allied Sciences
Colleges
Predictors
Resources
-
Law
Resources
Colleges
-
Animation and Design
Exams
Predictors & Articles
Colleges
Resources
-
Media, Mass Communication and Journalism
Colleges
Resources
-
Finance & Accounts
Top Courses & Careers
Colleges
- Home
- JEE Main 2020
- JEE Main 2020 Question Paper with Solution (Jan 8th second shift)
JEE Main 2020 Question Paper with Solution (Jan 8th second shift)
8th January, Afternoon Session ( 2:30 pm to 5:30 pm)
As we all know JEE main 2020 exam is conducted by the National test agency through online mode. The second day of the exam was the 8th of January which is held in two shifts Morning and Afternoon. As the JEE main 2020 exam on 8th January successfully conducted, aspirants may be eager to know the type of questions and level of exam. Through this article, you will get JEE main 2020 question paper with solution (Jan 8th second shift). Aspirants who appeared for the JEE main 2020 exam can check the solution and check their performance here.
Q1) What are the two monomers of maltose that are formed on hydrolysis?
Solution
The two monomers of maltose are:
-D-glucose and
-D-glucose
Q2) Which of the following is the correct order of the atomic radii of the elements C, O, F, Cl and Br?
Solution
The correct increasing order of the atomic radii of elements is given below:
F < O < C < Cl < Br
Atomic radii increase down a group and it decreases from left to right in a period.
Q3) Which of the following won't give Kjeldahl's test?
(a) C6H5NH2
(b) C6H5NO2
(c) NH2CONH2
(d) CH3CH2CN
Solution
Kjeldahl's method is not applicable to compounds containing nitrogen in nitro and azo compounds and nitrogen present in rings(pyridine) as nitrogen of these compounds does not convert to ammonium sulphate.
Therefore, Option(b) is correct.
Q4) Which of the following will show both frenkel and schottky defect?
1) AgBr
2) KBr
3) CaCl2
4) ZnS
Solution
Only AgBr can exhibit both Schottky and Frenkel defect.
Option 1 is correct
Q5) Arrange the different bonds according to their bond energies:
C-F, C-Cl, C-Br and C-I
Solution
The values of bond energies are given below:
C-F - 485kJ/mol
C-Cl - 328kJ/mol
C-Br - 276kJ/mol
C-I - 240kJ/mol
Thus, the correct order of bond energies is:
C-I > C-Br > C-Cl > C-F
Q6) Determine total number of neutrons in three isotopes of hydrogen.
(1) 1
(2) 2
(3) 3
(4) 4
Solution
Number of neutrons in Protium is 0.
Number of neutrons in Deuterium is 1.
Number of neutrons in Tritium is 2.
Thus, the correct answer is 0 + 1 + 2 = 3.
Q7) Determine Bohr's radius of Li2+ ion for n = 2. Given (Bohr's radius of H-atom = a0)
Solution
Option 2 is correct.
Q8) How many atoms lie in the same plane in the major product (C) ?
(Where A is the alkyne of lowest molecular mass)
Solution

Number of atoms in one plane = 13.
Q9) Complex [ML5] can exhibit trigonal bipyramidal and square pyramidal geometry. Determine the total number of 180º, 90º & 120º L-M-L bond angles.
Solution

Thus, the correct answer is 20.
Q10) Given the following reaction sequence

(1) Mg, Mg3N2
(2) Na, Na3N
(3) Mg, Mg(NO3)2
(4) Na, NaNO3
Solution
![]()
Option 1 is correct
Q11) Correct order of magnetic moment (spin only) for the following complexes
(a) [Pd(PPh3)2Cl2]
(b) [Ni(CO)4]
(c) [Ni(CN)4]2–
(d) [Ni(H2O)6]2+
(1) a = b = c < d
(2) a < b < c < d
(3) a > b > c > d
(4) a = b > c > d
Solution

Option 1 is correct.
Q12)

Compare Ea (activation energy) for a, b, c and d.
(1) Eb > Ec > Ed > Ea
(2) Ea > Ed > Ec > Eb
(3) Ec > Eb > Ea > Ed
(4) Ed > Ea > Eb > Ec
Solution
Eb > Ec > Ed > Ea
Option 1 is correct.
Q13) Given:
Basicity of B is:
(1) 1
(2) 2
(3) 3
(4) 4
Solution

Option 1 is correct.
Q14) Which reaction does not occurs in the blast furnace in the metallurgy of Fe

(1) a & b
(2) a,b & c
(3) c & d
(4) a, b, c, d
Solution
Concept Based
Option 3 is correct.
Q15) Correct order of radius of elements is: C, O, F, Cl, Br
(1) Br > Cl > C > O > F
(2) Br < Cl < C < O < F
(3) Cl < C < O < F < Br
(4) C > F > O > Br > Cl
Solution

Option 1 is correct
Q16) Amongs the following which will show geometrical isomerism.
(a) [Ni(NH3)5Cl]+
(b) [Ni(NH3)4ClBr]
(c) [Ni(NH3)3Cl]+
(d) [Ni(NH3)2(NO2)2]
(1) b, d
(2) a, b
(3) a, b & c
(4) a, b, c & d
Solution
Ma4bc can show 2 G.I.
Ma2b2 can show 2 G.I.
(Square planar)
Option 1 is correct.
Q17) Assertion: pH of water increases on increasing temperature.
Reason:
is an exothermic process.
(1) Both assertion and reason are correct and reason is correct explanation of assertion.
(2) Both assertion and reason are correct and reason is not correct explanation of assertion.
(3) Assertion is true & reason is false.
(4) Both assertion and reason are incorrect.
Solution
Concept Based.
Option 4 is correct.
Q18) Assertion: It has been found that for hydrogenation reaction the catalytic activity increases from group5 to group-11 metals with maximum activity being shown by groups 7-9 elements of the periodic table.
Reason: For 7-9 group elements adsorption rate is maximum.
(1) Both assertion and reason are correct and reason is correct explanation of assertion.
(2) Both assertion and reason are correct and reason is not correct explanation of assertion.
(3) Assertion is true & reason is false.
(4) Both are incorrect
Solution
Concept Based.
Option 1 is correct.
Q19) Given following reaction,
In the above reaction, 492 L of O2 is obtained at 1 atm & 300 K temperature. Determine mass of NaClO3 required (in kg). (R = 0.082 L atm mol–1 K–1)
Solution
moles of NaClO3 = moles of O2
Mass of NaClO3 = 20 x 106.5 = 2130g
Q20) Temperature of 4 moles of gas increases from 300 K to 500 K find 'Cv' if U = 5000 J.
Solution
U = nCv
T
5000 = 4 × Cv (500 – 300)
Cv = 6.25 JK–1 mol–1
Thus, the correct answer is 6.25
Q21) Formation of Bakelite follows:
(a) Electrophilic substitution followed by condensation.
(b) Nucleophilic addition followed by dehydration.
(c) Electrophilic addition followed by dehydration.
(d) Hydration followed by condensation.
Solution
Formation of Bakelite follows electrophilic substitution reaction of phenol with formaldehyde followed by condensation.


Q22) A compound X that adds 2 hydrogen molecules on hydrogenation. The compound X also gives 3-oxohexanedioic acid on oxidative ozonolysis. The compound 'X' is:

Solution

Q23) There are two compounds A and B of molecular formula C9H18O3. A has higher boiling point than B. What are the possible structures of A and B?

Solution
In (A), extensive inter-molecular H-bonding is possible while in (B) there is no Inter-molecular H-bonding.
Therefore, Option(2) is correct.
Q24) Find the final major product of the following reactions:

Solution

Therefore, Option(1) is correct
Q25) The major product of the following reactions is


Solution

Therefore, Option(3) is correct.
Q1. If a hyperbola has vertices (±6, 0) and P(10, 16) lies on it, then the equation of normal at P is
Solution:
Vertex is at (±6, 0)
a = 6
Let the hyperbola is
Putting point P(10, 16) on the hyperbola
Q2. How many 4 letter words can be formed with/without meaning using the word EXAMINATION?
Solution:
EXAMINATION 2N, 2A, 2I, E, X, M, T, O
Case I All are different so
Case II 2 same and 2 different so
Case III 2 same and 2 same so = 18
Total = 2454
Q3. In an A. P. 10th term is 1/20.and 20th term is 1/10.then find the sum of first 200th term
Solution:
Q4. The total number of observations is 20, the mean of the observation is 10 and the variance of the observation is 4 when taking incorrect reading 9 in place of 10. The correct variance is?
Solution:
Q5. Find the domain of the function having range (1,3)
Solution:
If x lies (1,2) [x]=1
Domain is
If x lies [2,3) [x]=2
Domain is .
Q6. Let is nonzero vector and
Solution:
Q7. Let the coefficient of and
in the expansion of
is
and
then
is equal to
Solution:
Q8. Differential equation of , where b is a parameter, is
Solution:
So the Differential equation is
Q9. Image of (1, 2, 3) w.r.t a plane is then which of the following points lie on the plane
Solution:
P(1,2,3) and Q
Direction ratio of normal to the plane
we get
and Midpoint of PQ is
equation of plane x + y + z = 1
Q10.
Solution:
Using L'Hospital
Q11. Let P be the set of points (x, y) such that . Then area of region bounded by points in set P is
Solution:
Point of intersection of is – 3, 1
Now,
Q12.
Solution:
Q13. Let the line y = mx intersects the curve at P and tangent to
at P intersects x-axis at Q. If area (
OPQ) = 4, find m (m > 0)
Solution:

Q14. Let where
Then tan (
) is equal to
Solution:
Q15. If y = mx + c is a tangent to the circle and also the perpendicular to the tangent to the circle
at
then
Solution:
The slope of the tangent to
so m = 1
y = x + c
now distance of (3, 0) from y = x + c is
Q16. Which of the following is a tautology
Solution:
Q17. Normal at (2, 2) to curve L. Then the perpendicular distance from the origin to line L is
Solution:
(2, 2) satisfy x – y = 0
Normal
x + y = = 4 Hence
the perpendicular distance from the origin
Q18. Let
Solution:
Q19. Let then value of
is
Solution:
On comparing we get
Q20. x + 2y + 2z = 5, 2
x + 3y + 5z = 8 and 4x +
y + 6z = 10 for the system of equation check the correct option.
(1) Infinite solutions when = 8 (2) Infinite solutions when
= 2
(3) no solutions when = 8 (4) no solutions when
=2
Solution:
D = ( + 8) ( 2 –
) for
= 2
= 5[18 – 10] – 2 [48 – 50] + 2 (16 – 30]
= 40 + 4 – 28 0 No solutions for
= 2
Q21. Let ,
If a and b are roots of quadratic equation then quadratic equation is ?
Solution:
=
, b = 1 +
3 +
6 + ……= 101
a= (1+) (1+
2+
4+
6.....
200)
x2 – 102x + 101 = 0
Q22. Let f(x) is a three degree polynomial for which f ' (–1) = 0, f '' (1) = 0, f(–1) = 10, f(1) = 6 then local minima of f(x) exist at
Solution:
Let f (x) = ax3 + bx2 + cx + d
a = 1/4, d = 35/4, b = –3/4, c =9/4
f(x) = a(x3 – 3x2 – 9x) + d
f ' (x) = 0 x = 3, –1
local minima exist at x = 3
Q23. Let A and B are two events such that then
Solution:
Q24. Solution set of contains
(1) singleton set
(2) two elements
(3) at least four elements
(4) infinite elements
Solution:

singolton set
Q-1) The efficiency of a carnot engine is 1/10. If this carnot engine is used as a refrigerator and the work required by it is 10J. Find the refrigeration effect of the refrigerator.
Sol-
Q-2 Find magnetic field at O.

Solution-

Magnitude of net magnetic field at O is
The direction of magnetic field due to first conductor ( in to the plane) is opposite to that of other two conductors (out of the plane).
Q 3- Position of particle as a function of time is given as . Choose correct statement about
where
are velocity and acceleration of particle at time t.
Options-
(1) is perpendicular to
and
is towards origin
(2) are perpendicular to
(3) is parallel to
and
parallel to
(4) is perpendicular to
and
is away from origin.
Sol-
implies a is anti parellel to r
Implies v is perpandicular to r
So option 1 is correct
Q 4- Two uniformly charged solid spheres are such that E1 is electric field at surface of 1st sphere due to itself. E2 is electric field at surface of 2nd sphere due to itself. r1, r2 are radius of 1st and 2nd sphere respectively. If then ratio of potential at the surface of spheres 1st and 2nd due to their self charges is:
Options:
sol-
Given
Q-5
Output at terminal Y of given logic circuit is

Options-
1) 0 2) 1 3) oscillating between o and 1 4) not determined
Sol-

Q 6- Velocity of a wave in a wire is v when tension in it is 2.06 × 104 N. Find value of tension in wire when velocity of wave become v/2.
Options:
(1) 5.15 × 103 N (2) 8.24 × 104 N (3) 6 × 104 (4) 5.15 × 104 N
Sol-
Velocity
Q 7- n mole of He and 2n mole of O2 is mixed in a container. Then will be
Options:
Sol-
For mono atomic gas (He)
For diatomic gas (O2)
Q-8 A uniform solid sphere of radius R has a cavity of radius 1m cut from it if centre of mass of the system lies at the periphery of the cavity then
Options
Sol-
Shift in centre of mass = 2-R
Q-9
A solid sphere of mass m= 500gm is rolling without slipping on a horizontal surface. Find kinetic energy of a sphere if velocity of centre of mass is 5 cm/sec.
Option:
Sol-
The kinetic energy of sphere is kinetic energy of translation+ kinetic energy of rotation
Where K is the radius of gyration
Q-10
Two liquid columns of same height 5m and densities and
are filled in a container of uniform cross sectional area. Then ratio of force exerted by the liquid on upper half of the wall to lower half of the wall.

Sol-

Q-11
Two square plates of side 'a' are arranged as shown in the figure. The minimum separation between plates is 'd' and one of the plate is inclined at small angle with plane parallel to another plate. The capacitance of capacitor is (given
is very small)

Solution-

Take an elemental strip of length dx at a distance x from one end
So the capacitance of an elemental strip is
So integrating this will give total capacitance=
(Using )
Q-12
In YDSE path difference at a point on screen is . Find ratio of intensity at this point with maximum intensity
Solution-
Intensity is given by
Where IO=maximum intensity
and using the Relation
we get
Q-13

Solution-
As current at any time t is given as
So
So integrating this will give total charge
Q-14
A particle is dropped from height h = 100 m, from surface of a planet. If in last (1/2) sec of its journey it covers 19 m. Then value of acceleration due to gravity that planet is :
Solution-
Let total time taken by particle is t
So distance covered by particle in t sec=
similarly distance covered by partical in sec=
So distance covered by partical in last (1/2) sec of its journey=
So
and
from equation (1) and (2)
we get
putting t=5 we get g = 8 m/s2
Q-15
A charged particle of mass m and charge q is released from rest in a uniform electric field. Its graph between velocity (v) and distance traveled (x) will be

Solution:-
So graph would be option 1
Q-16 An object is moving away from concave mirror of focal length f starting from focus. The distance of an object from pole of mirror is x. The correct graph of magnitude of magnification(m) verses distance x is:

Solution -
In this question we can use the elimination method by looking toward the graph.
As we know that the
And by using the mirror formula. we can write that the -
Now by using this equation, we can start eliminating the option. As we can see that the at u=f, the m become infinite. So the option (1) and (3) is incorrect. If we u = 2f, then the m =1.
So the option (3) is also wrong.
So the answer is option (4)
Q-17
In full scale deflection current in galvanometer of 100 resistance is 1 mA. Resistance required in series to convert it into voltmeter of range 10 V.
(1) 0.99 K (2) 9.9 K
(3) 9.8 K (4) 10 K
Soultion -
Q-18
There are two identical particles A and B. One is projected vertically upward with speed from ground and other is dropped from height h along the same vertical line. Collision between them is perfectly inelastic. Find time taken by them to reach the ground after collision in terms of
.
Solution -

time for collision -
After t1 -
and
at the time of collision
and height from ground
so time
So the option (1) is correct
Q-19
Length of a simple pendulum is 25.0 cm and time of 40 oscillation is 50 sec. If resolution of stop watch is 1 sec then accuracy is g is (in %)
(1) 2.4 (2) 3.4 (3) 4.4 (4) 5.4
Solution-
Note - We are adding the error just for calculating maximum possible error.
Q-20
Solution:-
We know v=u+at and here
So, magnitude of velocity:-
Q-21
An asteroid of mass is approaching with a velocity
when it is at distance of
from the centre of earth (where R is radius of earth). When it reaches at the surface of Earth, its velocity is (Nearest Integer) in km/s.
Solution:-
We will apply energy conservation here:-
Q-22
In H-spectrum wavelength of 1% line of Balmer series is . Find out wavelength of 2nd line of same series in nm.
Solution:-
Q-23
Solution:-
We know that:- and here s is same
Q-24
Solution:-

Q-25
Solution:-
Boost Your Preparation with these Knockout courses
Recently Asked Questions
-
Q.
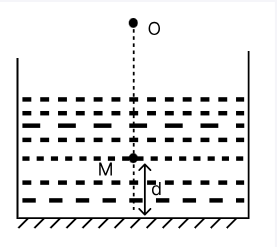
At the bottom of a tank having liquid with refractive index, 'm' is a plane mirror. A small object 'M' is situated at a height 'd' above the mirror. Object 'M' and its image in the mirror are visible to an observer who is 'O' vertically above M outside the liquid. The object 'M' and its image will appear to be separated by how much distance?
 Read More
Read More
-
Q.
A cubical volume is bounded by the surfaces
. The electric field in the region is given by
. Where
. If
, the charge contained in the cubical volume is
. The value of
is__________________.(Take
)
Option: 1288
Option: 2-
Option: 3-
Option: 4-
Read More
-
Q.
When a non-volatile solute is added to the solvent, the vapour pressure of the solvent decreases by 10 mm of Hg . The mole fraction of the solute in the solution is 0.2 . What would be the mole fraction of the solvent if the decrease in vapour pressure is 20 mm of Hg ?
1) (correct) 0.6
2) 0.4
3) 0.2
4) 0.8
Read More

