Browse by Stream
-
Engineering and Architecture
Exams
Colleges
Predictors
Resources
-
Computer Application and IT
Quick Links
Colleges
-
Pharmacy
Colleges
Resources
-
Hospitality and Tourism
Colleges
Resources
Diploma Colleges
-
Competition
Other Exams
Resources
-
School
Exams
Top Schools
Products & Resources
-
Study Abroad
Top Countries
Resources
-
Arts, Commerce & Sciences
Colleges
Upcoming Events
Resources
-
Management and Business Administration
Exams
Colleges & Courses
Predictors
-
Learn
Law Preparation
MBA Preparation
Engineering Preparation
Medical Preparation
-
Online Courses and Certifications
Top Streams
Specializations
- Digital Marketing Certification Courses
- Cyber Security Certification Courses
- Artificial Intelligence Certification Courses
- Business Analytics Certification Courses
- Data Science Certification Courses
- Cloud Computing Certification Courses
- Machine Learning Certification Courses
- View All Certification Courses
Resources
-
Medicine and Allied Sciences
Colleges
Predictors
Resources
-
Law
Resources
Colleges
-
Animation and Design
Exams
Predictors & Articles
Colleges
Resources
-
Media, Mass Communication and Journalism
Colleges
Resources
-
Finance & Accounts
Top Courses & Careers
Colleges
Get Answers to all your Questions


- Home
- Engineering
- The temperature of equal masses of three ifferent liquids x,y and z are <img alt="10^{\circ}C,20^{\circ}C \, and\, 30^{\circ}C" src="https://entrancecorner.codecogs.com/gif.latex?10%5E%7B%5Ccirc%7DC%2C20%5E%7B%5Ccirc%7DC%20%5C%2C%20and%5C%2C%2030%5E%7B%5C
The temperature of equal masses of three ifferent liquids x,y and z are 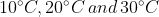 respectively.The temperature of mixture when x is mixed with y is
respectively.The temperature of mixture when x is mixed with y is 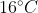 and that when y is mixed with z is
and that when y is mixed with z is 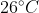 , The temperature of mixture when x and z are mixed will be :
, The temperature of mixture when x and z are mixed will be :
Option: 1 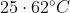
Option: 2 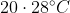
Option: 3 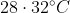
Option: 4 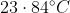
Answers (1)

For x & y mixture ,
Mixture of y & Z
Mixture of x & z
From eqn (1)
The correct option is (4)
View full answer
JEE Main high-scoring chapters and topics
Study 40% syllabus and score up to 100% marks in JEE


