Browse by Stream
-
Engineering and Architecture
Exams
Colleges
Predictors
Resources
-
Computer Application and IT
Quick Links
Colleges
-
Pharmacy
Colleges
Resources
-
Hospitality and Tourism
Colleges
Resources
Diploma Colleges
-
Competition
Other Exams
Resources
-
School
Exams
Top Schools
Products & Resources
-
Study Abroad
Top Countries
Resources
-
Arts, Commerce & Sciences
Colleges
Upcoming Events
Resources
-
Management and Business Administration
Exams
Colleges & Courses
Predictors
-
Learn
Law Preparation
MBA Preparation
Engineering Preparation
Medical Preparation
-
Online Courses and Certifications
Top Streams
Specializations
- Digital Marketing Certification Courses
- Cyber Security Certification Courses
- Artificial Intelligence Certification Courses
- Business Analytics Certification Courses
- Data Science Certification Courses
- Cloud Computing Certification Courses
- Machine Learning Certification Courses
- View All Certification Courses
Resources
-
Medicine and Allied Sciences
Colleges
Predictors
Resources
-
Law
Resources
Colleges
-
Animation and Design
Exams
Predictors & Articles
Colleges
Resources
-
Media, Mass Communication and Journalism
Colleges
Resources
-
Finance & Accounts
Top Courses & Careers
Colleges
Get Answers to all your Questions


- Home
- Engineering
- if a equation given and said to find oxidizing or reducing on what basis should we find ..like by ON or by electroN or addition of H
If an equation is given and said to find oxidizing or reducing. On what basis should we find.
Answers (1)
The various definitions of oxidation and reduction (redox) in terms of the transfer of oxygen, hydrogen, and electrons.
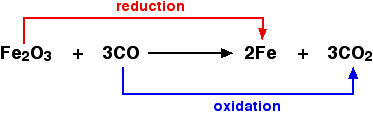
Oxidation and Reduction with respect to Oxygen Transfer
- Oxidation is the gain of oxygen.
- Reduction is the loss of oxygen.
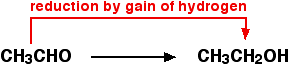
Oxidation and Reduction with respect to Hydrogen Transfer
- Oxidation is the loss of hydrogen.
- Reduction is the gain of hydrogen.
Notice that these are exactly the opposite of the oxygen definitions

Oxidation and Reduction with respect to Electron Transfer
- Oxidation is loss of electrons
- Reduction is gain of electrons
View full answer

