Browse by Stream
-
Engineering and Architecture
Exams
Colleges
Predictors
Resources
-
Computer Application and IT
Quick Links
Colleges
-
Pharmacy
Colleges
Resources
-
Hospitality and Tourism
Colleges
Resources
Diploma Colleges
-
Competition
Other Exams
Resources
-
School
Exams
Top Schools
Products & Resources
-
Study Abroad
Top Countries
Resources
-
Arts, Commerce & Sciences
Colleges
Upcoming Events
Resources
-
Management and Business Administration
Exams
Colleges & Courses
Predictors
-
Learn
Law Preparation
MBA Preparation
Engineering Preparation
Medical Preparation
-
Online Courses and Certifications
Top Streams
Specializations
- Digital Marketing Certification Courses
- Cyber Security Certification Courses
- Artificial Intelligence Certification Courses
- Business Analytics Certification Courses
- Data Science Certification Courses
- Cloud Computing Certification Courses
- Machine Learning Certification Courses
- View All Certification Courses
Resources
-
Medicine and Allied Sciences
Colleges
Predictors
Resources
-
Law
Resources
Colleges
-
Animation and Design
Exams
Predictors & Articles
Colleges
Resources
-
Media, Mass Communication and Journalism
Colleges
Resources
-
Finance & Accounts
Top Courses & Careers
Colleges
Get Answers to all your Questions


- #Chemistry
- #Chemical Bonding and Molecular Structure
- #NCERT
- #Chemistry Part I Textbook for Class XI
- #CBSE 11 Class
Although both are triatomic molecules, the shape of
molecule is bent while that of
is linear. Explain this based on the dipole moment.
Answers (1)
For regular structures or symmetrical structures, the dipole moment is 0. Unsymmetrical structures have a non-zero dipole moment. In the case of the net dipole moment is zero because the dipole of two equal bonds cancels with one another. The structure of carbon dioxide is-
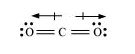 Resultant μ = 0 D
Resultant μ = 0 D
On the other hand dipole moment of is 1.84D.
is an unsymmetrical molecule and also bent geometries(not linear structure) because the bond polarities do not cancel each other. Therefore molecule
has a bent structure with a bond angle of 104°. The structure of water is-
 Resultant μ = 1.84 D
Resultant μ = 1.84 D
View full answer
Crack CUET with india's "Best Teachers"
- HD Video Lectures
- Unlimited Mock Tests
- Faculty Support

Similar Questions
- 0.3780 g of an organic chloro compound gave 0.5740 g of silver chloride in Carius estimation. Calculate the percentage of chlorine present in the compound.
- 1.0 mol of a monoatomic ideal gas is expanded from state (1) to state (2) as shown in figure. Calculate the work done for the expansion of gas from state (1) to state (2) at 298 K.
- At what temperature is the root mean square speed of an atom in an argon gas cylinder equal to the rms speed of a helium gas atom at – 20 °C ?


