Browse by Stream
-
Engineering and Architecture
Exams
Colleges
Predictors
Resources
-
Computer Application and IT
Quick Links
Colleges
-
Pharmacy
Colleges
Resources
-
Hospitality and Tourism
Colleges
Resources
Diploma Colleges
-
Competition
Other Exams
Resources
-
School
Exams
Top Schools
Products & Resources
-
Study Abroad
Top Countries
Resources
-
Arts, Commerce & Sciences
Colleges
Upcoming Events
Resources
-
Management and Business Administration
Exams
Colleges & Courses
Predictors
-
Learn
Law Preparation
MBA Preparation
Engineering Preparation
Medical Preparation
-
Online Courses and Certifications
Top Streams
Specializations
- Digital Marketing Certification Courses
- Cyber Security Certification Courses
- Artificial Intelligence Certification Courses
- Business Analytics Certification Courses
- Data Science Certification Courses
- Cloud Computing Certification Courses
- Machine Learning Certification Courses
- View All Certification Courses
Resources
-
Medicine and Allied Sciences
Colleges
Predictors
Resources
-
Law
Resources
Colleges
-
Animation and Design
Exams
Predictors & Articles
Colleges
Resources
-
Media, Mass Communication and Journalism
Colleges
Resources
-
Finance & Accounts
Top Courses & Careers
Colleges
Get Answers to all your Questions


(a) State the reason why carbon can neither form cations nor
anions, but forms covalent bonds. Also state reasons to explain why covalent compounds
(i) are bad conductors of electricity.
(ii) have low melting and boiling points.
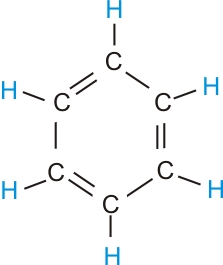
(b) Write the structural formula of benzene, .
Answers (1)
Atomic no. of carbon is . It means
electrons in its outermost shell.
- Carbon cannot form
cation, as very high energy is required to remove four valence electrons.
- Carbon cannot gain four electrons and form
anion as its nervous with 6 protons cannot hold
electrons.
(i) Covalent compounds are bad conductor of electricity because they do not have free electrons.
(ii) The intermolecular forces of attraction between the covalent compounds are weak, so, they have quite low melting and boiling point.
(b) Structure of Benzene.
View full answer


