Browse by Stream
-
Engineering and Architecture
Exams
Colleges
Predictors
Resources
-
Computer Application and IT
Quick Links
Colleges
-
Pharmacy
Colleges
Resources
-
Hospitality and Tourism
Colleges
Resources
Diploma Colleges
-
Competition
Other Exams
Resources
-
School
Exams
Top Schools
Products & Resources
-
Study Abroad
Top Countries
Resources
-
Arts, Commerce & Sciences
Colleges
Upcoming Events
Resources
-
Management and Business Administration
Exams
Colleges & Courses
Predictors
-
Learn
Law Preparation
MBA Preparation
Engineering Preparation
Medical Preparation
-
Online Courses and Certifications
Top Streams
Specializations
- Digital Marketing Certification Courses
- Cyber Security Certification Courses
- Artificial Intelligence Certification Courses
- Business Analytics Certification Courses
- Data Science Certification Courses
- Cloud Computing Certification Courses
- Machine Learning Certification Courses
- View All Certification Courses
Resources
-
Medicine and Allied Sciences
Colleges
Predictors
Resources
-
Law
Resources
Colleges
-
Animation and Design
Exams
Predictors & Articles
Colleges
Resources
-
Media, Mass Communication and Journalism
Colleges
Resources
-
Finance & Accounts
Top Courses & Careers
Colleges
Get Answers to all your Questions


- #Class 11
- #National Eligibility Cum Entrance Test
- #National Eligilibility Cum Entrance Test
- #Chemistry
- #Medical
- #Hydrogen
At absolute zero temperature:
Option: 1
Only para hydrogen exists
Option: 2
Only ortho hydrogen exist
Option: 3
Both para and ortho hydrogen exist
Option: 4
None
Answers (1)

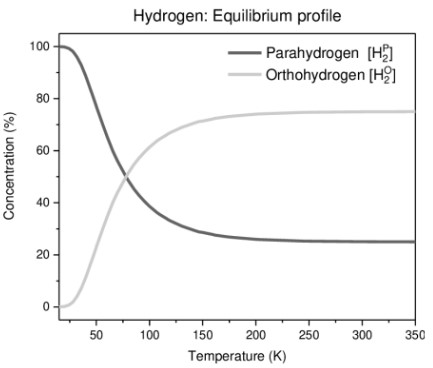
A molecule of dihydrogen contains two atoms. The nuclei of both the atoms in each molecule of dihydrogen are spinning. Depending upon the direction of the spins of the nuclei, hydrogen is of two types - Ortho hydrogen and Para hydrogen.

Ordinary dihydrogen is an equilibrium mixture of ortho and para hydrogen. Para hydrogen is more stable than ortho hydrogen at lower temperature. Hence at 0 K, hydrogen exist only as para hydrogen. Concentration of Ortho and para hydrogen with temperature is given hereunder:
View full answer
NEET 2024 Most scoring concepts
- Just Study 32% of the NEET syllabus and Score up to 100% marks


